PriceList for Chemiluminescence Immunoassay - C-creactive protein CRP rapid test kit – Baysen
PriceList for Chemiluminescence Immunoassay - C-creactive protein CRP rapid test kit – Baysen Detail:
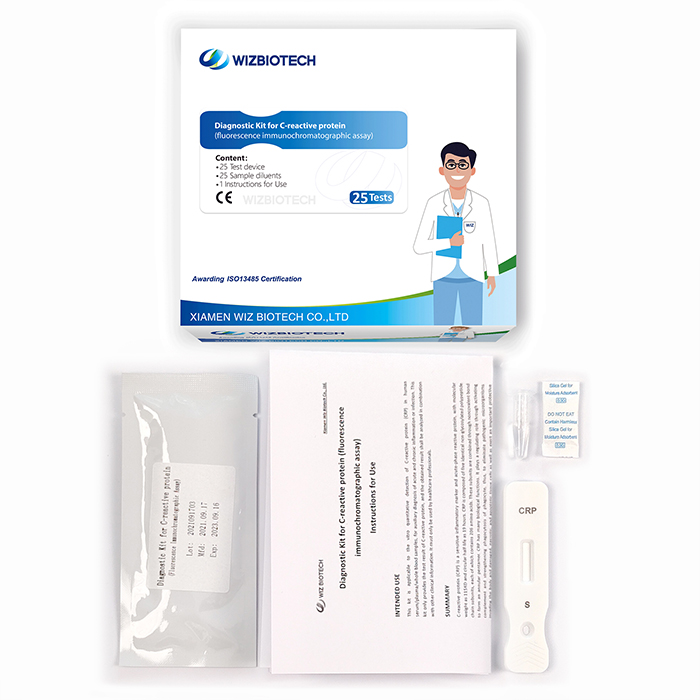
Diagnostic Kit for C-reactive Protein
Methodology:Fluorescence Immunochromatographic Assay
Production information
| Model Number | CRP | Packing | 25 Tests/ kit, 30kits/CTN |
| Name | Diagnostic Kit for
C-reactive Protein |
Instrument classification | Class I |
| Features | High sensitivity, Easy opeation | Certificate | CE/ ISO13485 |
| Accuracy | > 99% | Shelf life | Two Years |
| Methodology | Fluorescence Immunochromatographic Assay | OEM/ODM service | Avaliable |
INTENDED USE
This kit is applicable to the vitro quantitative detection of C-reactive protein (CRP) in human serum/plasma/whole blood samples, for auxiliary diagnosis of acute and chronic inflammation or infection. This kit only provides the test result of C-reactive protein, and the obtained result shall be analyzed in combination with other clinical information.
Test procedure
| 1 | Before using the reagent,read the package insert carefully and familiarize yourself with the operating procedures. |
| 2 | Select standard test mode of WIZ-A101 portable immune analyzer |
| 3 | Open the aluminum foil bag package of reagent and take out the test device. |
| 4 | Horizontally insert the test device into the slot of immune analyzer. |
| 5 | On home page of operation interface of immune analyzer, click “Standard” to enter test interface. |
| 6 | Click “QC Scan” to scan the QR code on inner side of the kit; input kit related parameters into instrument and select sample type. Note: Each batch number of the kit shall be scanned for one time. If the batch number has been scanned, then skip this step. |
| 7 | Check the consistency of “Product Name”, “Batch Number” etc. on test interface with information on the kit label. |
| 8 | Take out sample diluent upon consistent information, add 10μL serum/plasma/whole blood sample, and thoroughly mix them; |
| 9 | Add 80µL aforesaid thoroughly mixed solution into well of test device; |
| 10 | After complete sample addition, click “Timing” and remaining test time will be automatically displayed on the interface. |
| 11 | Immune analyzer will automatically complete test and analysis when test time is reached. |
| 12 | After test by immune analyzer is completed, test result will be displayed on test interface or can be viewedthrough “History” on home page of operation interface. |
Note: each sample shall be pipetted by clean disposable pipette to avoid cross contamination.
Product detail pictures:


Related Product Guide:
Alleviating the agony and the angst of prostate cancer over-diagnosis Australian Financial Review Australian Financial Review | P24 Test Strips
Commutability of the First World Health Organization International Standard for Human Cytomegalovirus | Cpn-Igm
We are experienced manufacturer. Wining the majority from the crucial certifications of its market for PriceList for Chemiluminescence Immunoassay - C-creactive protein CRP rapid test kit – Baysen , The product will supply to all over the world, such as: Belarus, Lithuania, Argentina, Many goods fully conform to the most rigorous of international guidelines and with our first-rate delivery service you will have them delivered at any time and in any place. And because Kayo deals in the entire spectrum of protective equipment, our customers don't have to waste time shopping around.
It is really lucky to meet such a good supplier, this is our most satisfied cooperation, I think we will work again!