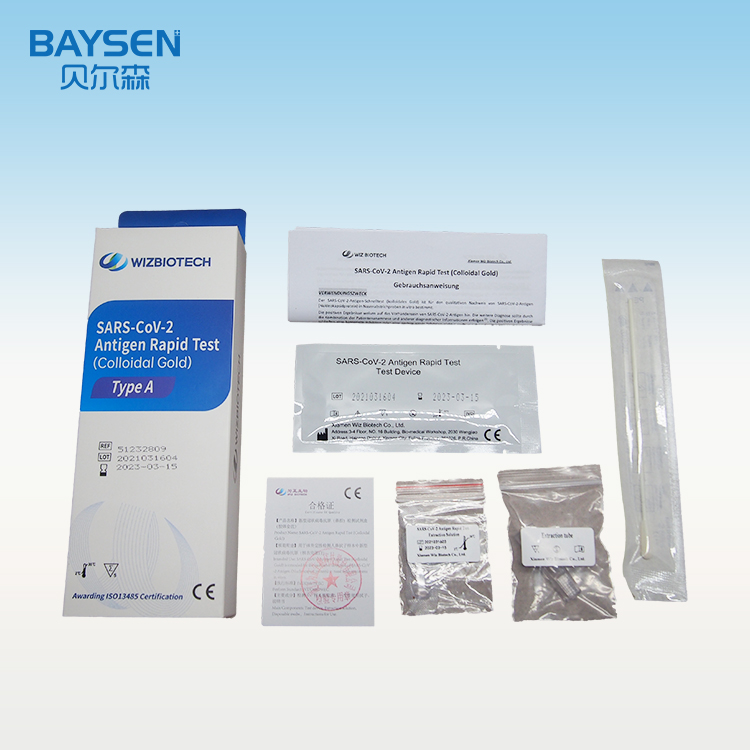
Manufacturing Companies for Urine Analysis Strip - Diagnostic Kit for Antibody to Helicobacter Pylori(Fluorescence Immunochromatographic Assay) – Baysen
Manufacturing Companies for Urine Analysis Strip - Diagnostic Kit for Antibody to Helicobacter Pylori(Fluorescence Immunochromatographic Assay) – Baysen Detail:
Diagnostic Kit for Antibody to Helicobacter Pylori(Fluorescence Immunochromatographic Assay)
For in vitro diagnostic use only
Please read this package insert carefully prior to use and strictly follow the instructions. Reliability of assay results cannot be guaranteed if there are any deviations from the instructions in this package insert.
INTENDED USE
Diagnostic Kit for Antibody to Helicobacter Pylori (Fluorescence Immunochromatographic Assay)is a fluorescence immunochromatographic assay for the quantitative detection of HP antibody in human serum or plasma. which is important auxiliary diagnostic value for gastric infections.All positive sample must be confirmed by other methodologies. This test is intended for healthcare professional use only.
SUMMARY
Gastric helicobacter pylori infection is closely related with chronic gastritis, gastric ulcer, gastric adenocarcinoma, gastric mucosa associated lymphoma, H.p ylori infection rate of about 90% in gastritis, gastric ulcer, duodenal ulcer and gastric cancer patients. WHO has identified h. pylori as the first type of cancer-causing factor.it is a risk factor for gastric cancer.H.pylori detection is of great value in the diagnosis of h. pylori infection.
PRINCIPLE OF THE PROCEDURE
The membrane of the test device is coated with HP antigen on the test region and goat anti rabbit IgG antibody on the control region. Lable pad are coated by fluorescence labeled HP antigen and rabbit IgG in advance. When testing positive sample, the HP antibody in sample combine with fluorescence labeled HP antigen, and form immune mixture. Under the action of the immunochromatography, the complex flow in the direction of absorbent paper, when complex passed the test region, it combined with HP coating antigen, forms new complex.HP-Ab level is positively correlated with fluorescence signal, and the concentration of HP-Ab in sample can be detected by fluorescence immunoassay assay
REAGENTS AND MATERIALS SUPPLIED
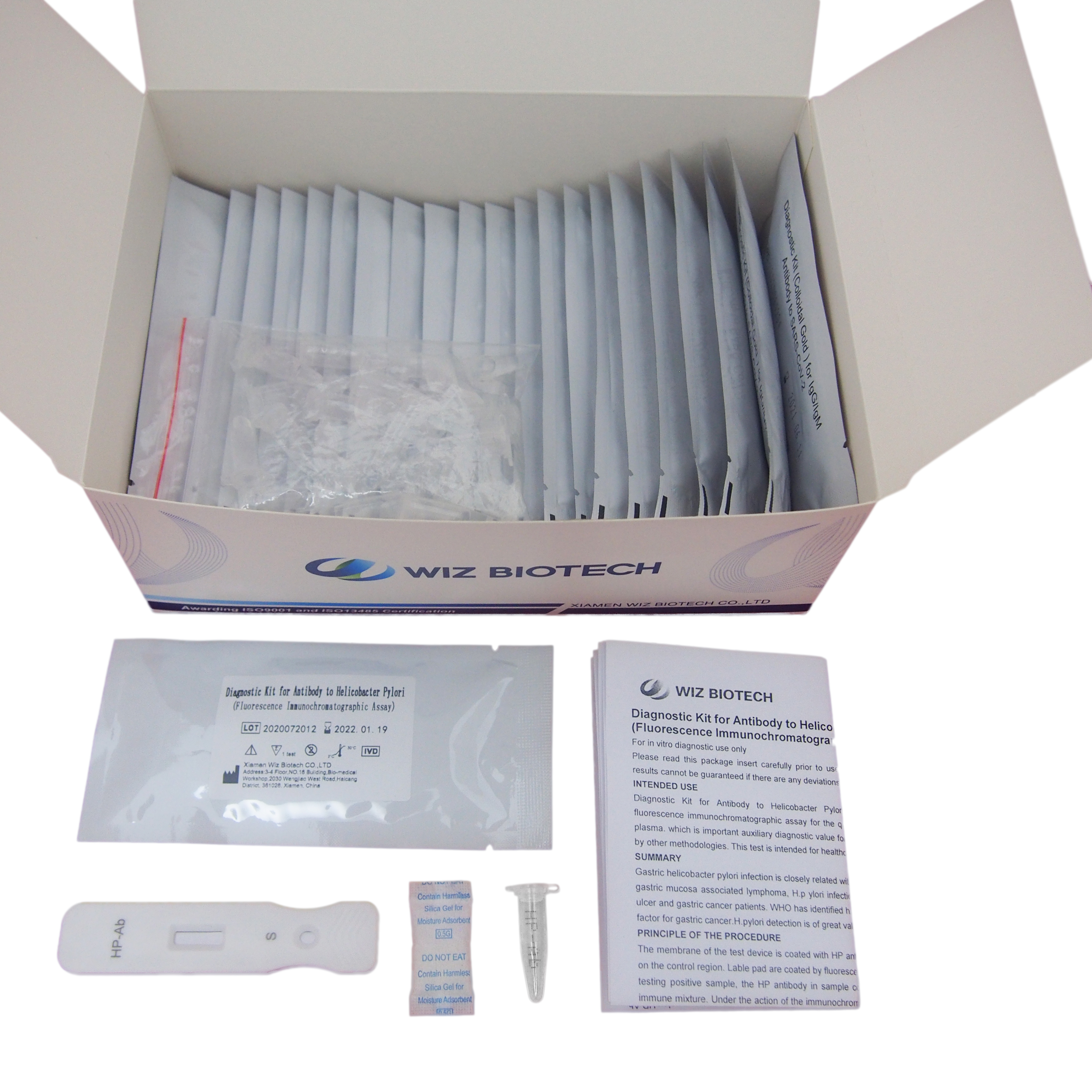
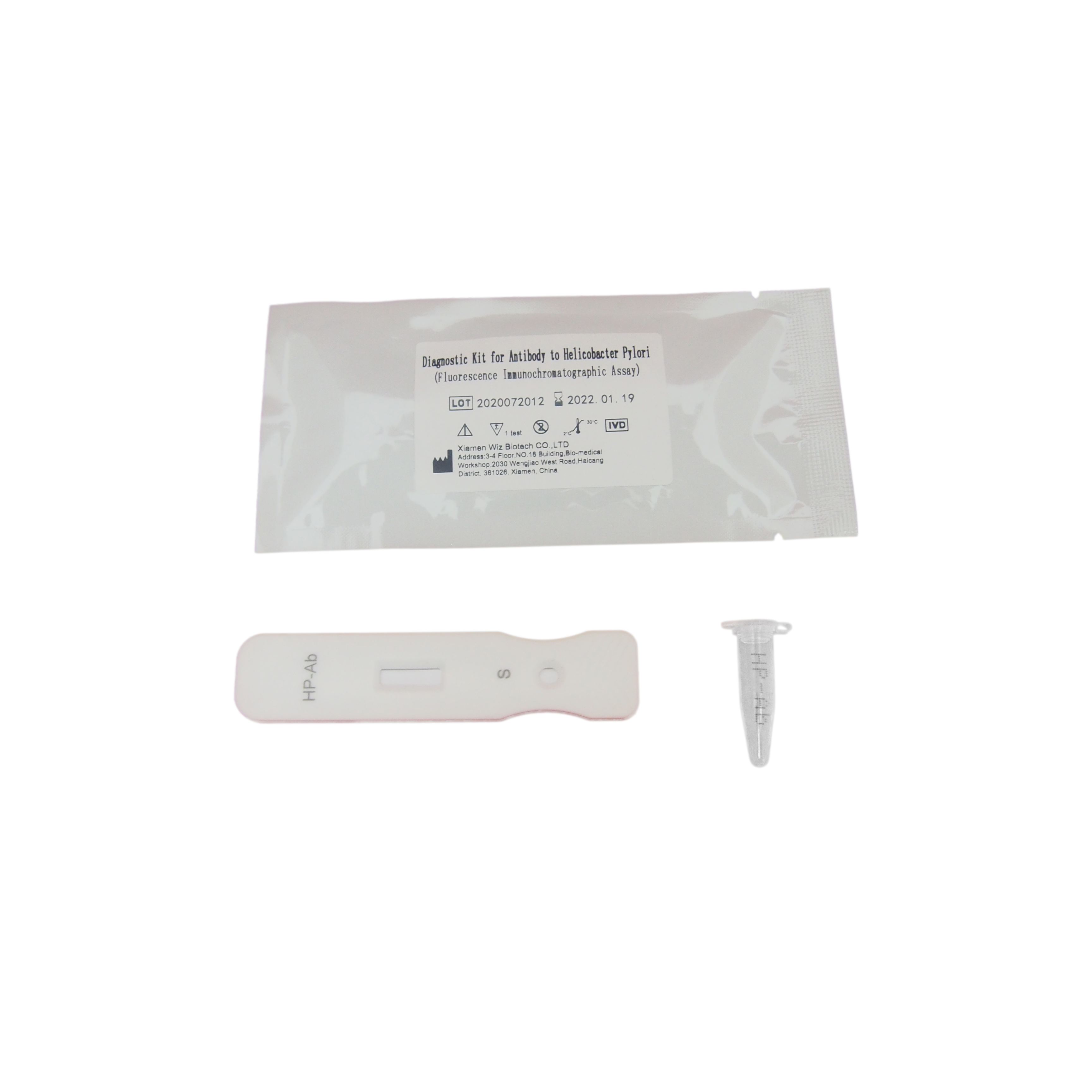
25T package components:
Test card individually foil pouched with a desiccant 25T
Sample diluents 25T
Package insert 1
MATERIALS REQUIRED BUT NOT PROVIDED
Sample collection container,timer
SAMPLE COLLECTION AND STORAGE
1.The samples tested can be serum, heparin anticoagulant plasma or EDTA anticoagulant plasma.
2.According to standard techniques collect sample. Serum or plasma sample can be kept refrigerated at 2-8℃ for 7days and cryopreservation below -15°C for 6 months.
3.All sample avoid freeze-thaw cycles.
ASSAY PROCEDURE
Please read the instrument operation manual and package insert before testing.
1.Lay aside all reagents and samples to room temperature.
2.Open the Portable Immune Analyzer(WIZ-A101), enter the account password login according to the operation method of the instrument, and enter the detection interface.
3.Scan the dentification code to confirm the test item.
4.Take out the test card from the foil bag.
5.Insert the test card into the card slot, scan the QR code, and determine the test item.
6.Add 20μL serum or plasma sample to sample diluent, and mix well..
7.Add 80μL sample solution to sample well of the card.
8.Click the “standard test” button, after 15 minutes, the instrument will automatically detect the test card, it can read the results from the display screen of the instrument, and record/print the test results.
9.Refer to the instruction of Portable Immune Analyzer(WIZ-A101).
EXPECTED VALUES
HP-Ab<10
It is recommended that each laboratory establish its own normal range representing its patient population.
TEST RESULTS AND INTERPRETATION
.The above data is the result of HP-Ab reagent test, and it is suggested that each laboratory should establish a range of HP-Ab detection values suitable for the population in this region. The above results are for reference only.
.The results of this method are only applicable to the reference ranges established in this method, and there is no direct comparability with other methods.
.Other factors can also cause errors in detection results, including technical reasons, operational errors and other sample factors.
STORAGE AND STABILITY
1.The kit is 18 months shelf-life from the date of manufacture. Store the unused kits at 2-30°C. DO NOT FREEZE. Do not use beyond the expiration date.
2.Do not open the sealed pouch until you are ready to perform a test, and the single-use test is suggested to be used under the required environment (temperature 2-35℃, humidity 40-90%) within 60 mins as quickly as possible.
3.Sample diluent is used immediately after being opened.
WARNINGS AND PRECAUTIONS
.The kit should be sealed and protected against moisture.
.All positive specimens shall be validated by other methodologies.
.All specimens shall be treated as potential pollutant.
.DO NOT use expired reagent.
.DO NOT interchange reagents among kits with different lot No..
.DO NOT reuse test cards and any disposable accessories.
.Misoperation, excessive or little sample can lead to result deviations.
LIMITATION
.As with any assay employing mouse antibodies, the possibility exists for interference by human anti-mouse antibodies (HAMA) in the specimen. Specimens from patients who have received preparations of monoclonal antibodies for diagnosis or therapy may contain HAMA. Such specimens may cause false positive or false negative results.
.This test result is only for clinical reference, should not serve as the only basis for clinical diagnosis and treatment, the patients clinical management should be comprehensive consideration combined with its symptoms, medical history, other laboratory examination, treatment response, epidemiology and other information.
.This reagent is only used for serum and plasma tests. It may not obtain accurate result when used for other samples such as saliva and urine and etc.
PERFORMANCE CHARACTERISTICS
| Linearity | 10-1000 | relative deviation:-15% to +15%. |
| Linear correlation coefficient:(r)≥0.9900 | ||
| Accuracy | The recovery rate shall be within 85% – 115%. | |
| Repeatability | CV≤15% | |
REFERENCES
1.Shao,J.L.&F.Wu.Recent advances in the detection methods of Helicobacter pylori[J].Journal of Gastroenterology and Hepatology,2012,21(8):691-694
2.Hansen JH,et al.HAMA Interference with Murine Monoclonal Antibody-Based Immunoassays[J].J of Clin Immunoassay,1993,16:294-299.
3.Levinson SS.The Nature of Heterophilic Antibodies and the Role in Immunoassay Interference[J].J of Clin Immunoassay,1992,15:108-114.
Key to symbols used:
 |
In Vitro Diagnostic Medical Device |
 |
Manufacturer |
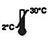 |
Store at 2-30℃ |
 |
Expiration Date |
 |
Do Not Reuse |
 |
CAUTION |
 |
Consult Instructions For Use |
Xiamen Wiz Biotech CO.,LTD
Address:3-4 Floor,NO.16 Building,Bio-medical Workshop,2030 Wengjiao West Road,Haicang District,361026,Xiamen,China
Tel:+86-592-6808278
Fax:+86-592-6808279
Product detail pictures:
Related Product Guide:
It’s time for a new Europe-wide strategy on prostate cancer | Psa Test Cost
Een kudtkoekiewall. Omdat dat moet, van de kudtkoekiewet. | P24 Test Strips
By using a full scientific excellent administration method, great quality and fantastic religion, we get good reputation and occupied this discipline for Manufacturing Companies for Urine Analysis Strip - Diagnostic Kit for Antibody to Helicobacter Pylori(Fluorescence Immunochromatographic Assay) – Baysen , The product will supply to all over the world, such as: Mozambique, United Kingdom, Jakarta, To get more information about us as well as see all our products, please visit our website. To get more information please feel free to let us know. Thank you very much and wish your business always be great!
Goods just received, we are very satisfied, a very good supplier, hope to make persistent efforts to do better.