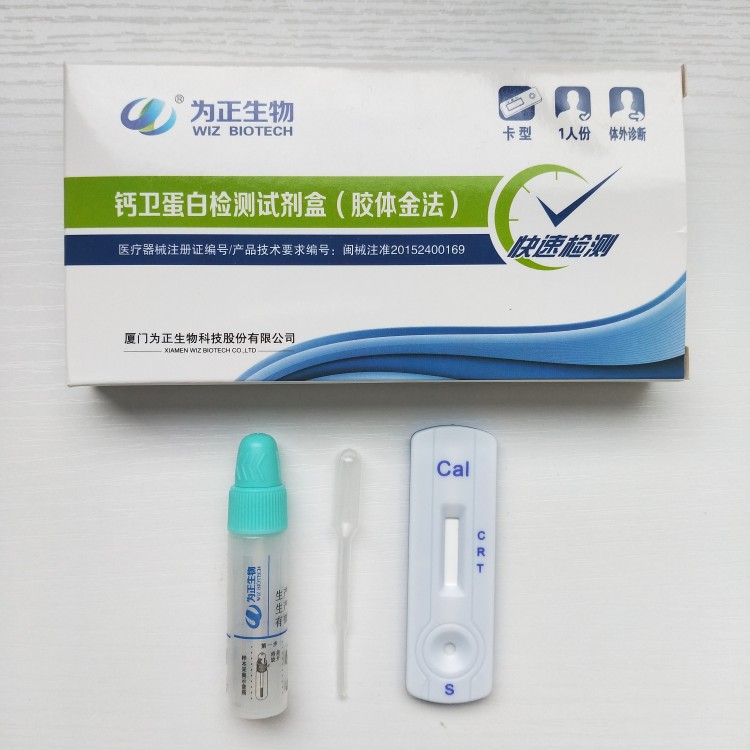
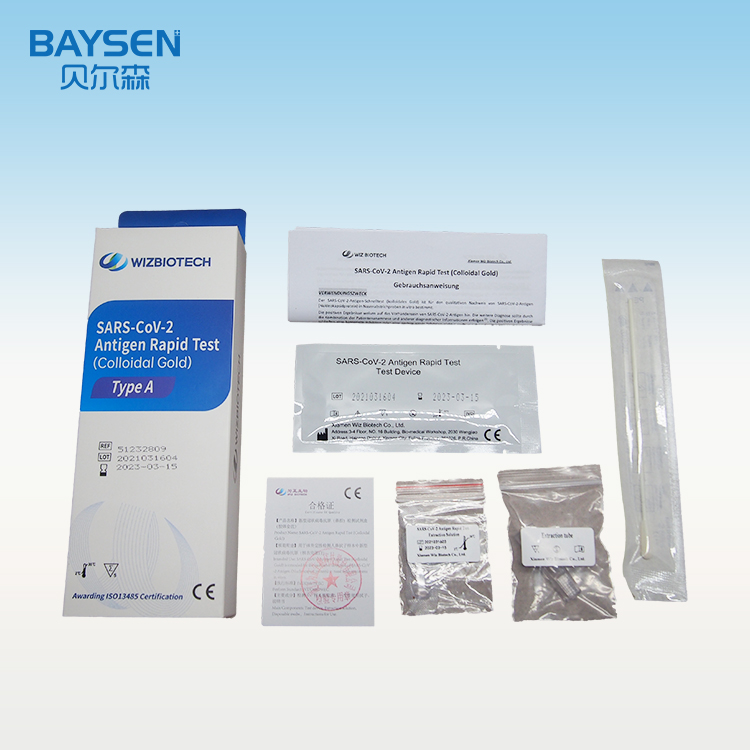
Manufactur standard Ce And Fda Approved Lh Test - Diagnostic kit for Carcino-embryonic antigen ( Fluorescence immunochromatographic assay) – Baysen
Manufactur standard Ce And Fda Approved Lh Test - Diagnostic kit for Carcino-embryonic antigen ( Fluorescence immunochromatographic assay) – Baysen Detail:
| Products | Specifications | code | unit | linear range | reference range | Linear range |
| Diagnostic kit for Carcino-embryonic antigen | 25T/Box | Cea | ng/ml | 2-500 | <5 |
Product detail pictures:



Related Product Guide:
Direct Reading of Bona Fide Barcode Assays for Diagnostics with Smartphone Apps | Diagnostic Kit For Isoenzyme Mb Of C Reatine Kinase
Metabolic Signatures of Exercise in Human Plasma | P24 Test Strips
To create more value for customers is our business philosophy; customer growing is our working chase for Manufactur standard Ce And Fda Approved Lh Test - Diagnostic kit for Carcino-embryonic antigen ( Fluorescence immunochromatographic assay) – Baysen , The product will supply to all over the world, such as: Barcelona, Niger, Denmark, We adhere to client 1st, top quality 1st, continuous improvement, mutual advantage and win-win principles. When cooperation together with the customer, we provide shoppers with the highest high-quality of service. Established good business relations using the Zimbabwe buyer inside the business, we've got established own brand and reputation. At the identical time, wholeheartedly welcome new and old prospects to our company to go to and negotiate small business.
It can be said that this is a best producer we encountered in China in this industry, we feel lucky to work with so excellent manufacturer.