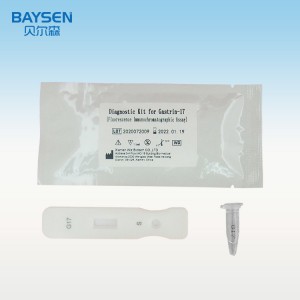
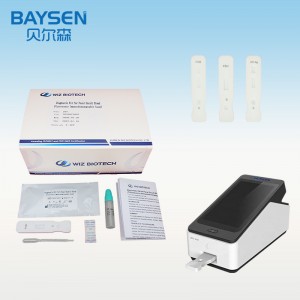
Instrumentum diagnosticum ad usum in grosso Gastrin-17 POCT, reagens detectionis rapidae
Ut simus scena somniorum operariorum nostrorum implendorum! Ut laetiorem, multo unitiorem et multo peritiorem turmam aedificemus! Ut commodum mutuum clientium nostrorum, suppeditorum, societatis et nostrum pro apparatu diagnostico Gastrin-17 POCT celeris detectionis reagenti assequamur. Novos et senes clientes ex omnibus vitae generibus libenter invitamus ut nos ad futuras societates negotiales et mutuas res gestas vocent.
Ut simus locus ubi somnia operariorum nostrorum impleantur! Ut laetiorem, multo unitiorem et multo peritiorem gregem aedificemus! Ut commodum mutuum clientium nostrorum, praebitorum, societatis et nostrum assequamur.Instrumentum Probationis Sinensis, Supplementa MedicaCum qualitate optima, pretio modico, traditione tempore suo, et officiis ad usum aptatis ad clientes perveniendum, societas nostra laudem et in mercatibus domesticis et externis accepit. Emptores nobiscum contactum facere possunt.
Libellus FOB
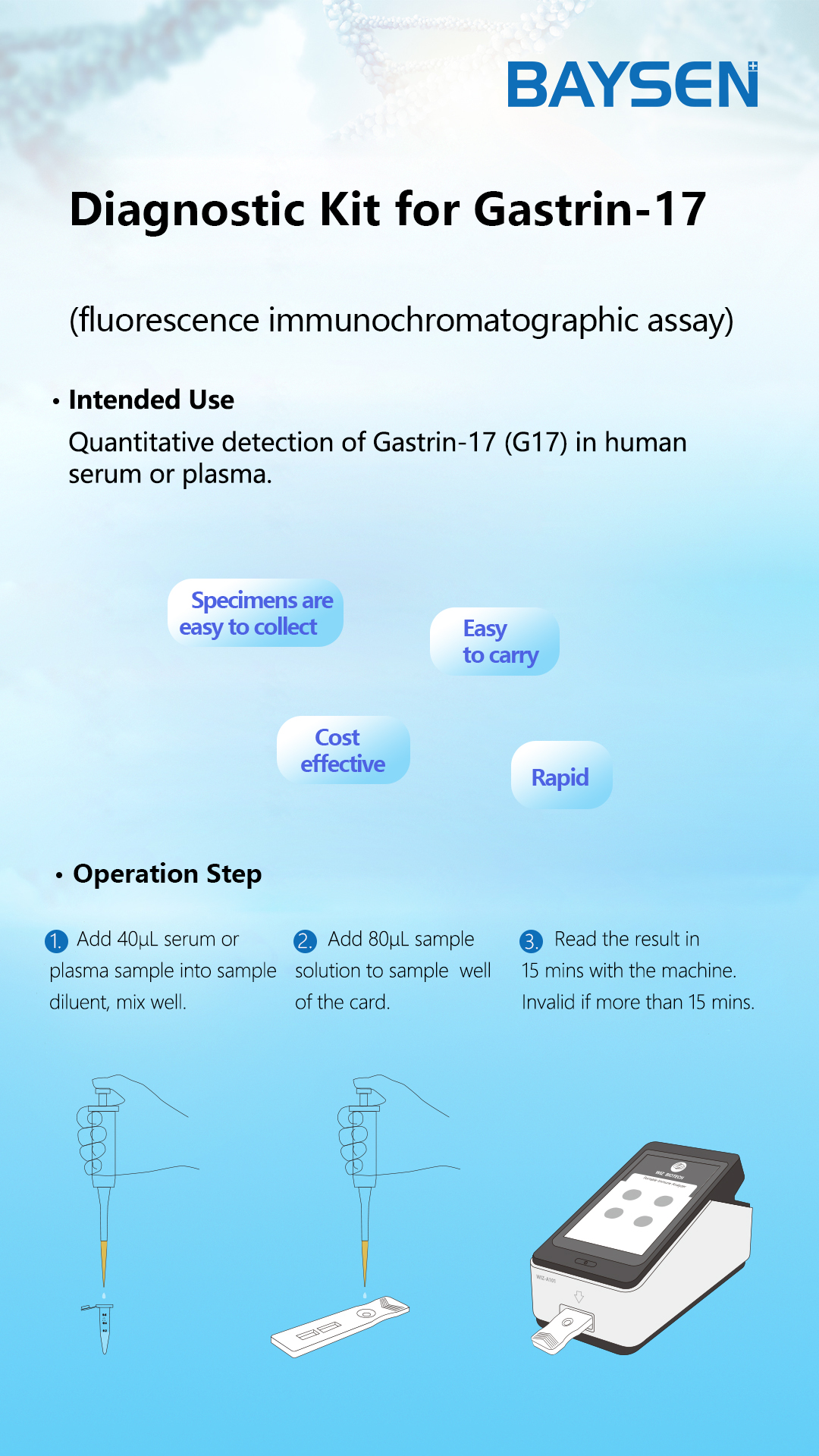

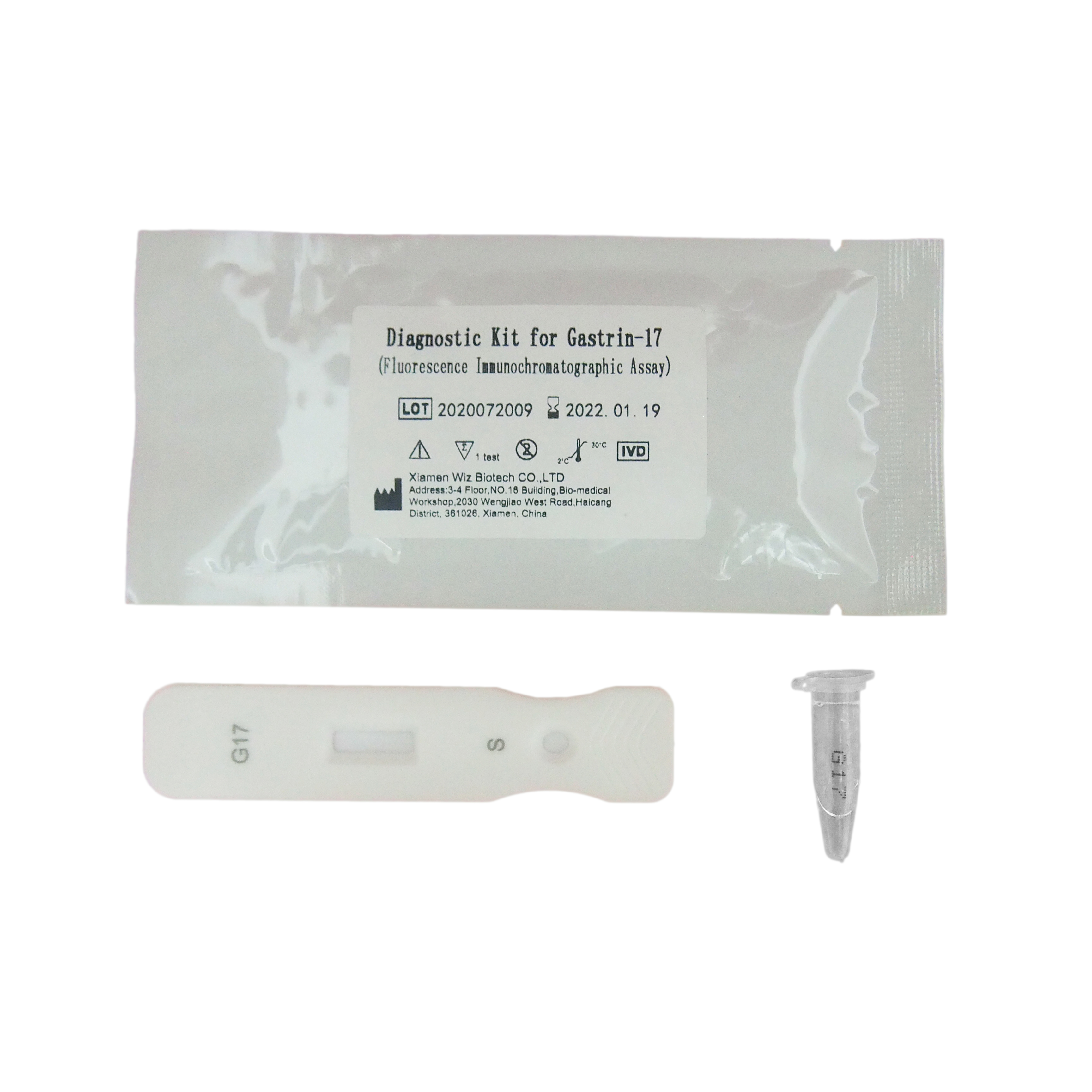
Principium et Ratio Probationis FOB
Principium:
Taenia anticorpus anti-FOB in regione probationis habet, quod antea chromatographiae membranae affixum est. Tegumentum inscriptio anticorpore anti-FOB fluorescentia notato antea obducitur. Cum exemplum positivum examinatur, FOB in exemplo cum anticorpore anti-FOB fluorescentia notato misceri potest, et mixturam immunem formare. Dum mixtura per taeniam migrare permittitur, complexus FOB coniugatus ab anticorpore anti-FOB in membrana capitur et complexum format. Intensitas fluorescentiae positive cum contento FOB correlata est. FOB in exemplo per analysatorem immunologicum fluorescentiae detegi potest.
Modus Probationis:
1. Omnia reagentia et exempla ad temperaturam cubiculi seponantur.
2. Aperi Analysorem Immunitatis Portatilem (WIZ-A101), inscribe tesseram inscriptionis secundum modum operationis instrumenti, et intra interfaciem detectionis.
3. Codicem identificationis inspice ut rem probatam confirmes.
4. Chartam probationis e sacculo aluminio extrahe.
5. Chartam probationis in fissuram chartae inser, codicem QR lege, et rem probatam determina.
6. Operculum e tubo speciminis remove et primas duas guttas speciminis diluti abice. Tres guttas (circiter 100µL) speciminis diluti sine bullis verticaliter et lente in puteum speciminis chartae cum dispense incluso adde.
7. Preme bullam "probationis ordinariae"; post XV minuta, instrumentum chartam probationis sponte deteget, eventus ex monitorio instrumenti legere, et eventus probationis notare/imprimere potest.
8. Instructiones Analysatoris Immunitatis Portatilis (WIZ-A101) vide.

Fortasse tibi placebit
Examen Celere Antigeni SARS-CoV-2 (Aurum Colloidale)
WIZ-A101 Analysator Immunitatis Portatilis
Instrumentum Diagnosticum Sanguinis Occulti Faecalis (Examen Immunochromatographicum Fluorescens)
De Nobis

Xiamen Baysen Medical Tech Limited est societas biologica summae artis quae se in campo reagentium diagnosticorum celerium dedicat et investigationem et progressionem, productionem et venditionem in unum integrum integrat. Multi sunt in societate investigatores provecti et rectores venditionum, omnes qui amplam experientiam laboris in Sinis et societatibus biopharmaceuticis internationalibus habent.
Exhibitio certificati