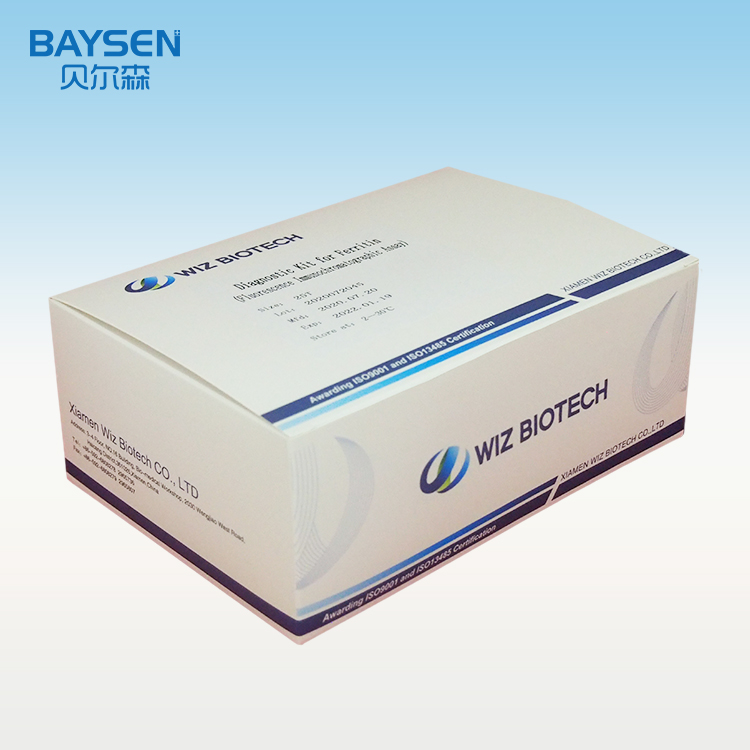
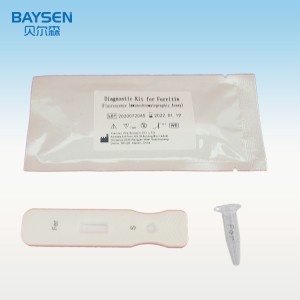
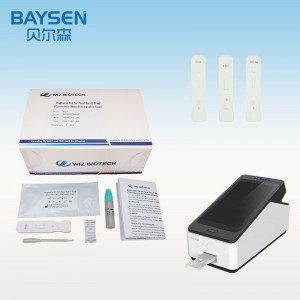
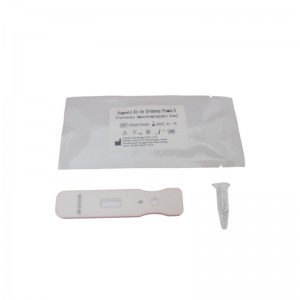
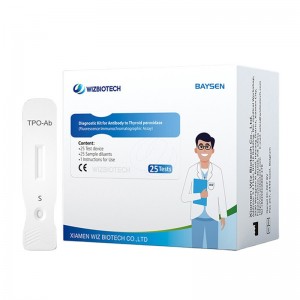
Officina OEM pro Sinis, probatione rapida, alta accuratio, alta qualitate, charta integra, HIV, HBV, HBsag, HCV, Dengue, Malaria, Tp, Syphilis, Fob, Fer, Zika, Influenza, Ab, H5, H7.
Progressus inculcamus et novas merces in forum singulis annis introducimus pro Officina OEM pro Sinis Rapid Test High Accuracy High Quality Uncut Sheet HIV HBV HBsag HCV Dengue Malaria Tp Syphilis Fob Fer Zika Influenza Ab H5 H7. Sincere speramus nos cum emptoribus nostris toto orbe terrarum crescere.
Progressum inculcamus et novas merces in forum singulis annis introducimus.Examen Chartae Magnae Sinensis, Examen Chartae IntegræCum de ullis rebus nostris, postquam indicem productorum nostrorum inspexeris, delectaberis, libenter nobiscum contactum fac si quaestiones habes. Epistulas electronicas nobis mittere et nobiscum ad consultationem communicare potes, et quam primum tibi respondebimus. Si tibi commodum erit, inscriptionem nostram in situ nostro interretiali invenire et ad officinam nostram venire potes, vel plura de rebus nostris ipse scire. Semper parati sumus ad diuturnam et stabilem cooperationem cum quibusvis clientibus potentialibus in campis conexis construendam.
Parametri Productorum

Principium et Ratio Probationis FOB
PRINCIPIUM
Membrana instrumenti probationis anticorpore anti FER in regione probationis et anticorpore caprino anti FER IgG in regione moderatrice obducta est. Laminae inscriptionales anticorpore anti FER fluorescentia notato et IgG leporino antea obducuntur. Cum exemplum positivum probatur, antigenum FER in exemplo cum anticorpore anti FER fluorescentia notato miscetur, mixturam immunologicam formans. Sub actione immunochromatographiae, complexus in directionem chartae absorbentis fluit; cum complexus regionem probationis transit, cum anticorpore anti FER obducto miscetur, novum complexum formans. Gradus FER positive cum signo fluorescentiae correlatus est, et concentratio FER in exemplo per immunoassay fluorescentiae detegi potest.
Ratio Probationis
Quaeso, antequam experimentum facias, manuale operationis instrumenti et insertum involucri perlege.
1. Omnia reagentia et exempla ad temperaturam cubiculi seponantur.
2. Aperi Analysorem Immunitatis Portatilem (WIZ-A101), inscribe tesseram inscriptionis secundum modum operationis instrumenti, et intra interfaciem detectionis.
3. Codicem identificationis lege ut rem probatam confirmes.
4. Chartam probationis e sacculo aluminio extrahe.
5. Chartam probationis in fissuram chartae inser, codicem QR lege, et rem probatam determina.
6. Adde 40μL seri vel plasmatis speciminis in diluentem speciminis, et bene misce.
7. 80μL solutionis speciminis in puteum speciminis chartae adde.
8. Preme bullam "probationis ordinariae"; post XV minuta, instrumentum chartam probationis sponte deteget, eventus ex monitorio instrumenti legere, et eventus probationis notare/imprimere potest.
9. Instructiones Analysatoris Immunitatis Portatilis (WIZ-A101) vide.

Fortasse tibi placebit
Examen Celere Antigeni SARS-CoV-2 (Aurum Colloidale)
WIZ-A101 Analysator Immunitatis Portatilis
Instrumentum Diagnosticum pro 25-hydroxy Vitamino D (Examen Immunochromatographicum Fluorescens)
De Nobis

Xiamen Baysen Medical Tech Limited est societas biologica summae artis quae se in campo reagentium diagnosticorum celerium dedicat et investigationem et progressionem, productionem et venditionem in unum integrum integrat. Multi sunt in societate investigatores provecti et rectores venditionum, omnes qui amplam experientiam laboris in Sinis et societatibus biopharmaceuticis internationalibus habent.
Exhibitio certificati