Instrumentum Diagnosticum pro Myoglobino Troponino I Cardiaco et Isoenzymo MB Creatini Kinasis
Instrumentum Diagnosticum Troponini I Cardiaci ∕Isoenzymi MB Creatini Kinasis ∕Myoglobini
Methodologia: Examen Immunochromatographicum Fluorescens
Informationes de productione
| Numerus Modeli | cTnI/CK-MB/MYO | Sarcinatio | XXV probationes/sarcina, XXX sarcinae/CTN |
| Nomen | Instrumentum Diagnosticum Troponini I Cardiaci ∕Isoenzymi MB Creatini Kinasis ∕Myoglobini | Classificatio instrumentorum | Classis II |
| Proprietates | Alta sensibilitas, facilis operatio | Certificatum | CE/ ISO13485 |
| Accuratio | > 99% | Tempus conservationis | Biennium |
| Methodologia | Examen Immunochromatographicum Fluorescentiae | Officium OEM/ODM | Disponibile |
USUS INTENTUS
Hoc instrumentum ad detectionem quantitativam in vitro concentrationum signorum laesionis myocardialis cardiacorum applicatur.
troponinum I, isoenzymum MB creatini kinasini et myoglobinum in sero/plasma/sanguine humano, et
Aptum est ad diagnosim auxiliarem infarctus myocardii. Hoc instrumentum tantummodo probationes troponini I cardiaci praebet.
isoenzyma MB creatini kinaseini et myoglobini, et eventus obtenti in combinatione cum aliis adhibebuntur.
Informationes clinicae ad analysin. Solum a peritis medicis adhibendae sunt.
Modus probationis
| 1 | Antequam reagente utaris, diligenter lege insertum involucri et te cum rationibus operandi familiariza. |
| 2 | Modum probationis ordinarium analysatoris immunitatis portatilis WIZ-A101 elige. |
| 3 | Sacculum aluminii reagentis e charta aperi et instrumentum probationis extrahe. |
| 4 | Instrumentum probationis horizontaliter in fissuram analysatoris immunitatis inseratur. |
| 5 | In pagina prima interfaciei operationis analysatoris immunitatis, preme "Standard" ad interfaciem probationis ingrediendum. |
| 6 | Preme "QC Scan" ut codicem QR in latere interiore apparati perlegas; parametros apparati pertinentes in instrumentum inscribe et genus exempli elige. Nota: Numerus quisque partis apparati semel perscrutandus est. Si numerus partis perscrutatus est, hunc gradum omitte. |
| 7 | Congruentiam "Nominis Producti", "Numeri Seriei" etc. in interfacie probationis cum informatione in pittacio apparati verifica. |
| 8 | Diluens speciminis exime, informatione constans observata, 80 μL seri/plasmatis/sanguinis totius adde, et diligenter misce; |
| 9 | Adde 80µL solutionis praedictae perfecte mixtae in puteum instrumenti probationis; |
| 10 | Post additionem exemplaris completam, "Tempus" preme et tempus probationis reliquum in interfacie automatice demonstrabitur. |
| 11 | Analysator immunitatis probationem et analysin automatice perficiet cum tempus probationis advenerit. |
| 12 | Postquam probatio per analysatorem immunologicum perfecta est, eventus probationis in interfacie probationis demonstrabitur vel per "Historiam" in pagina prima interfaciei operationis videri potest. |
Nota: singula exempla pipetta munda et abicienda perfundenda sunt ne contaminatio mutua fiat.
Superioritas
Tempus probationis: 10-15 minuta
Conservatio: 2-30℃/36-86℉
Methodologia: Examen Immunochromatographicum Fluorescens
Proprietas:
• Alta sensibilitas
• Lectio eventus intra XV minutas
• Facilis operatio
• Tres probationes uno tempore, tempora conservantes.
• Alta Praecisio
Effectus Clinicus
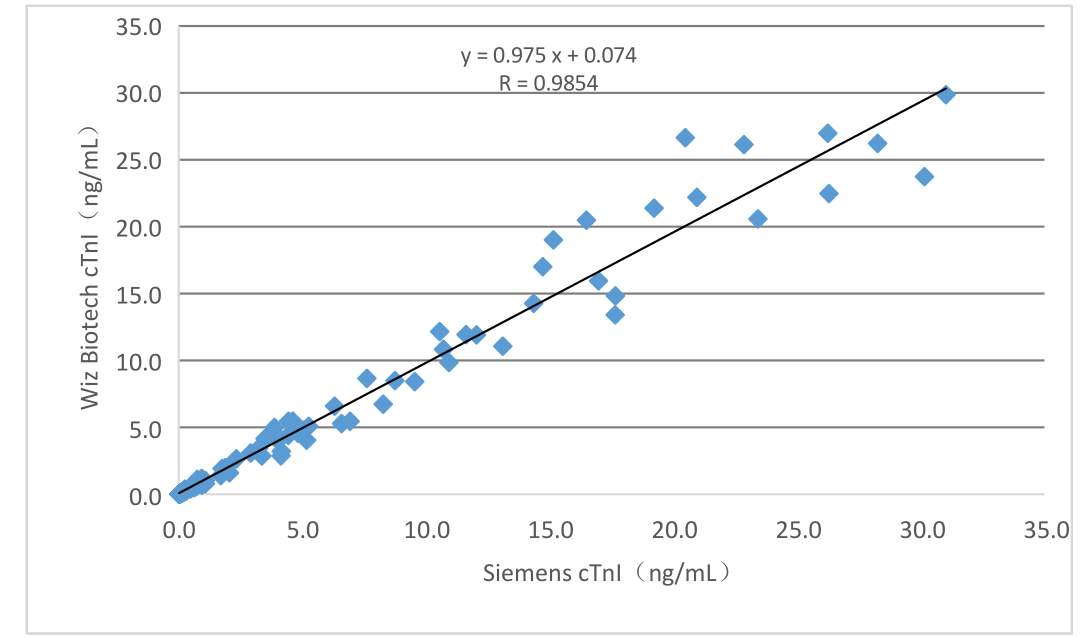
Effectus clinicus huius producti per collectionem 150 casuum exemplorum clinicorum aestimatus est.
a) In casu medicamenti cTnI, apparatus correspondens venalis probationum chemiluminescentiae ut reagens referentiale adhibitum,
Resultata detectionis comparata sunt et comparabilitas eorum per regressionem linearem investigata est, et
Coefficientes correlationis duarum probationum sunt Y = 0.975X + 0.074 et R = 0.9854 respective;
b) In casu rei CK-MB, apparatus correspondens venalis probationum electrochemiluminescentiae ut referentia adhibitus.
reagens, eventus detectionis comparati sunt et eorum comparabilitas per linearem investigata est.
regressio, et coefficientes correlationis duorum experimentorum sunt Y = 0.915X + 0.242 et R = 0.9885 respective.
c) In casu rei MYO, apparatus correspondens venalis probationum immunologicarum fluoris tempore resolutarum ut referentia adhibitus.
reagens, eventus detectionis comparati sunt et eorum comparabilitas per linearem investigata est.
regressio, et coefficientes correlationis duarum probationum sunt y = 0.989x + 2.759 et R = 0.9897 respective.
Fortasse etiam tibi placebunt: