High Quality for Pathology Reagent Test Kits Cpn Igm - Diagnostic Kit(Colloidal Gold)for Follicle-stimulating Hormone – Baysen
High Quality for Pathology Reagent Test Kits Cpn Igm - Diagnostic Kit(Colloidal Gold)for Follicle-stimulating Hormone – Baysen Detail:
Diagnostic Kit(Colloidal Gold)for Follicle-stimulating Hormone
For in vitro diagnostic use only
Please read this package insert carefully prior to use and strictly follow the instructions. Reliability of assay results cannot be guaranteed if there are any deviations from the instructions in this package insert.
INTENDED USE
The kit is used for qualitative detection of follicle-stimulating hormone (FSH) levels in urine samples. It is suitable for assisting determination the appearance of female menopause.
PACKAGE SIZE
1 kit /box, 10 kits /box, 25 kits,/box, 50 kits /box.
SUMMARY
FSH is a glycoprotein hormone secreted by the pituitary gland, it can enter blood and urine through blood circulation. For male, FSH promotes maturity of testicular seminiferous tubule and the production of sperm, for female, FSH promotes follicular development and maturity, and collaborates LH to mature follicles secrete estrogen and ovulation, involved in the formation of normal menstruation[1]. FSH maintains a consistently stable basal level in normal subjects, about 5-20mIU/mL. Female menopause typically occurs between the ages of 49 and 54, and lasts for an average of four to five years. During this period, because of ovarian atrophy, follicular atresia and degeneration, estrogen secretion decreased significantly, a large number of stimulating pituitary gonadotropin secretion, especially FSH levels will be significantly increased, is generally 40-200mIU/ml, and maintain the level in a very long time[2]. This kit based on colloidal gold immune chromatography analysis technology for qualitative detection of FSH antigen in human urine samples, which can give a result within 15 minutes.
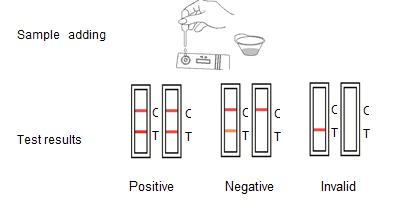
ASSAY PROCEDURE
1.Take out the test card from the foil bag, put it on the level table and mark it.
2.Discard the first two drops sample, add 3 drops (about 100μL) no bubble sample verticaly and slowly into sample well of the card with provided dispette, start timing.
3.The result should be read within 10-15 minutes, and it is invalid after 15 minutes.
Product detail pictures:

Related Product Guide:
Should PSA test be free for all men in B.C.? | Diagnostic Kit For Isoenzyme Mb Of C Reatine Kinase
Most Men Don’t Need a PSA Test for Prostate Cancer | Psa Test Cost
No matter new shopper or old customer, We believe in very long expression and dependable relationship for High Quality for Pathology Reagent Test Kits Cpn Igm - Diagnostic Kit(Colloidal Gold)for Follicle-stimulating Hormone – Baysen , The product will supply to all over the world, such as: Colombia, Greece, Indonesia, Our company now has many department, and there have more than 20 employees in our company. We set up sales shop, show room, and product warehouse. In the meantime, we registered our own brand. We've got tightened inspection for quality of product.
We have been engaged in this industry for many years, we appreciate the work attitude and production capacity of the company, this is a reputable and professional manufacturer.