Factory best selling Dengue Ns1 Fever Rapid Test Kit - Diagnostic Kit(Colloidal Gold)for IgM Antibody to Human Enterovirus 71 – Baysen
Factory best selling Dengue Ns1 Fever Rapid Test Kit - Diagnostic Kit(Colloidal Gold)for IgM Antibody to Human Enterovirus 71 – Baysen Detail:
Diagnostic Kit(Colloidal Gold)for IgM Antibody to Human Enterovirus 71
For in vitro diagnostic use only
Please read this package insert carefully prior to use and strictly follow the instructions. Reliability of assay results cannot be guaranteed if there are any deviations from the instructions in this package insert.
INTENDED USE
Diagnostic Kit(Colloidal Gold)for IgM Antibody to Human Enterovirus 71 is a colloidal gold immunochromatographic assay for the qualitative determination of IgM Antibody to Human Human Enterovirus 71(EV71-IgM) in human whole blood,serum or plasma.This test is a screening reagent. All positive sample must be confirmed by other methodologies.This test is intended for healthcare professional use only.
PACKAGE SIZE
1 kit /box, 10 kits /box, 25 kits,/box, 50 kits /box
Summary
EV71 is one of the main pathogens of hand, foot and mouth disease(HFMD), which can cause myocarditis, encephalitis,acute respiratory disease and other diseases except HFMD. The Kit is a simple, visual qualitative test that detects EV71-IgM in human whole blood, serum or plasma. The Diagnostic Kit is based on immunochromatography and can give a result within 15 minutes.
Applicable instrument
Except visual inspection, the kit can be matched with Continuous immune analyzer WIZ-A202 of Xiamen Wiz Biotech Co., Ltd
ASSAY PROCEDURE
The WIZ-A202 test procedure see the instruction of Continuous immune analyzer. Visual test procedure is as follows
1.Take out the test card from the foil bag, put it on the level table and mark it.
2.Add 10μl serum or plasma sample or 20ul whole blood sample to sample well of the card with provided dispette, then add 100μl (about 2-3 drop) sample diluent; start timing
3.Wait for a minimum 10-15 minutes and read the result, the result is invalid after 15 minutes.
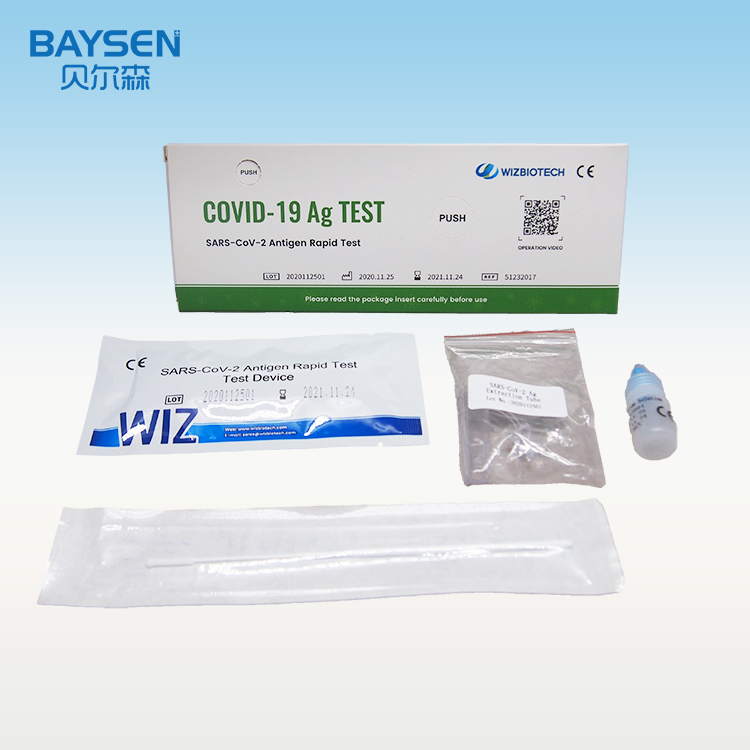
Product detail pictures:

Related Product Guide:
Ces études à la con qui nous prennent pour des connes | P24 Test Strips
Identification and Clinical Significance of Helcococcus species, with Description of Helcococcus seattlensis sp. nov. from a Patient with Urosepsis | Diagnostic Kit For Isoenzyme Mb Of C Reatine Kinase
With our leading technology as well as our spirit of innovation,mutual cooperation, benefits and development, we will build a prosperous future together with your esteemed company for Factory best selling Dengue Ns1 Fever Rapid Test Kit - Diagnostic Kit(Colloidal Gold)for IgM Antibody to Human Enterovirus 71 – Baysen , The product will supply to all over the world, such as: Paris, Jakarta, Mecca, We have customers from more than 20 countries and our reputation has been recognized by our esteemed customers. Never-ending improvement and striving for 0% deficiency are our two main quality policies. Should you need anything, don't hesitate to contact us.
The factory technical staff gave us a lot of good advice in the cooperation process, this is very good, we are very grateful.